|
Description
|
|
Patient Condition
- A 80-year-old female patient with acute cholangitis.
- The patient was in preparation for general anesthesia.
Events
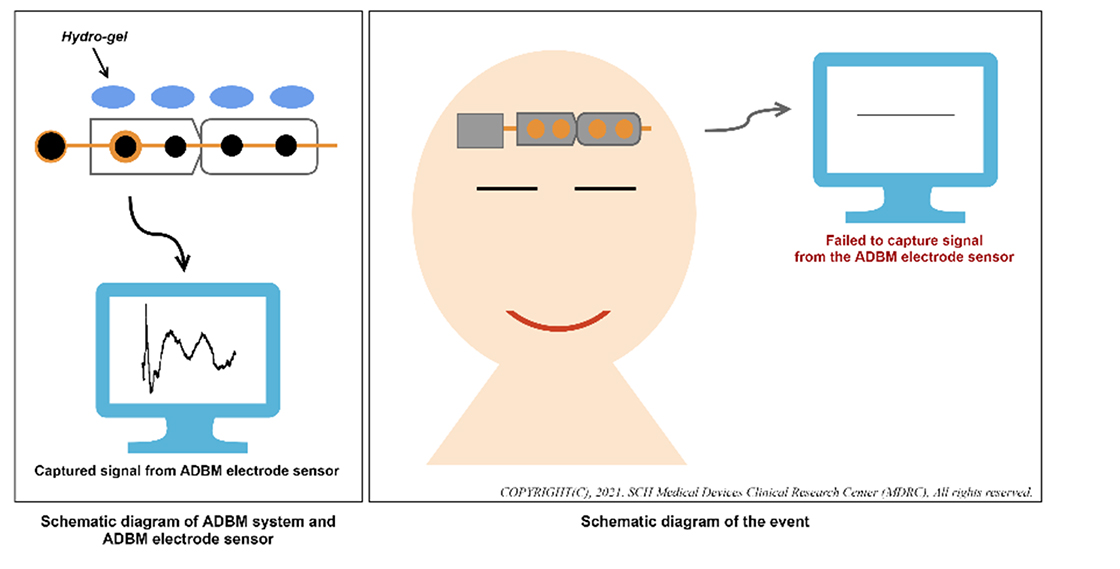
- Before starting general anesthesia, the ADBM electrode sensor (the sensor) was applied to the patient’s forehead.
- The ADBM system failed to display the EEG signal on its monitor.
- The nurse confirmed that the expiration date of the sensor had not passed.
Post-event management & Health effect
- The electrode sensor was replaced with another sensor of the same lot and it worked properly.
- Nurses are experiencing frequent sensor detection errors, especially in older patients. They assume that the skin condition may have an impact, so particular attention is payed when attaching the sensor to elderly patients.
Investigations (Cause, Improvements)
- The quality manager of the manufacturer received the problematic sensor from the user.
- When the problematic sensor was tested by connecting it to an ADBM system by the manufacturer, the system detected the signal normally.
- The quality manager checked the hydro-gel status of other products in the same lot and found that it conformed to the manufacturer's manufacturing standards.
|
|
Click to Zoom
|
|
Annex
|
Code
|
#
|
%
|
| A |
A01 - Lower Level Codes
| A01 |
Patient Device Interaction Problem |
15 |
|
| A0101 |
Patient-Device Incompatibility |
4 |
|
|
19
|
31.1%
|
| A |
A07 - Lower Level Codes
| A07 |
Electrical /Electronic Property Problem |
2 |
|
| A0701 |
Capturing Problem |
1 |
|
| A070908 |
Failure to Sense |
1 |
|
| A0710 |
Failure to Conduct |
1 |
|
|
5
|
8.2%
|
| A |
A26 - Lower Level Codes
| A26 |
Insufficient Information |
5 |
|
|
5
|
8.2%
|
| A |
A12 - Lower Level Codes
|
5
|
8.2%
|
| A |
A22 - Lower Level Codes
| A22 |
Human-Device Interface Problem |
2 |
|
| A2201 |
Device Difficult to Setup or Prepare |
1 |
|
| A2203 |
Device Difficult to Maintain |
1 |
|
|
4
|
6.6%
|
| A |
A02 , A04 , A11 , A13 , A03 , A05 , A09 , A17 , A21 , A23 , A24 , A25 |
23
total:61
|
37.7%
|
| B |
B01 - Lower Level Codes
| B01 |
Testing of Actual/Suspected Device |
23 |
|
|
23 |
50.0%
|
| B |
B02 - Lower Level Codes
| B02 |
Testing of Device from Same Lot/Batch Retained by Manufacturer |
8 |
|
|
8 |
17.4%
|
| B |
B03 - Lower Level Codes
| B03 |
Testing of Device from Same Lot/Batch Returned from User |
7 |
|
|
7 |
15.2%
|
| B |
B07 - Lower Level Codes
| B07 |
Testing of Raw/Starting Materials |
2 |
|
|
2 |
4.3%
|
| B |
B05 - Lower Level Codes
| B05 |
Testing of Device from Other Lot/Batch Returned From User |
1 |
|
|
1 |
2.2%
|
| B |
B09 , B14 , B21 , B22 , B04 |
5
total:46 |
10.9%
|
| C |
C19 - Lower Level Codes
| C19 |
No Device Problem Found |
12 |
|
| 12 |
25.5%
|
| C |
C01 - Lower Level Codes
| C01 |
Biological Problem Identified |
6 |
|
| 6 |
12.8%
|
| C |
C02 - Lower Level Codes
| C02 |
Electrical Problem Identified |
6 |
|
| 6 |
12.8%
|
| C |
C23 - Lower Level Codes
| C23 |
Usage Problem Identified |
4 |
|
| 4 |
8.5%
|
| C |
C14 - Lower Level Codes
| C14 |
Patient Sample Problem |
2 |
|
| C1402 |
Known Interferent |
1 |
|
| 3 |
6.4%
|
| C |
C03 , C04 , C13 , C06 , C07 , C09 , C10 , C16 , C20 , C21 , C05 |
16
total:47 |
34.0%
|
| D |
D14 - Lower Level Codes
| D14 |
No Problem Detected |
9 |
|
| 9 |
23.1%
|
| D |
D01 - Lower Level Codes
| D01 |
Cause Traced to Device Design |
5 |
|
| 5 |
12.8%
|
| D |
D11 - Lower Level Codes
| D11 |
Cause Traced to User |
2 |
|
| D1102 |
Unintended Use Error Caused or Contributed to Event |
1 |
|
| D1104 |
Shelf Life/Expiration Date Exceeded |
2 |
|
| 5 |
12.8%
|
| D |
D10 - Lower Level Codes
| D10 |
Cause Traced to Non-Device Related Factors |
3 |
|
| D1001 |
Adverse Event Related to Patient Condition |
1 |
|
| 4 |
10.3%
|
| D |
D15 - Lower Level Codes
| D15 |
Cause Not Established |
3 |
|
| 3 |
7.7%
|
| D |
D16 , D02 , D06 , D08 , D17 , D04 , D07 , D13 |
13
total:39 |
33.3%
|
| E |
E24 - Lower Level Codes
| E24 |
Others |
3 |
|
| E2403 |
No Clinical Signs, Symptoms or Conditions |
12 |
|
| 15 |
38.5%
|
| E |
E01 - Lower Level Codes
| 7 |
17.9%
|
| E |
E22 - Lower Level Codes
| E22 |
Investigations and Diagnostic Tests |
3 |
|
| 3 |
7.7%
|
| E |
E17 - Lower Level Codes
| E17 |
Skin and Subcutaneous Tissue |
3 |
|
| 3 |
7.7%
|
| E |
E21 - Lower Level Codes
| E21 |
Procedural Complications |
1 |
|
| E2102 |
Awareness during Anaesthesia |
1 |
|
| 2 |
5.1%
|
| E |
E08 , E11 , E23 , E02 , E03 , E04 , E06 |
9
total:39 |
23.1%
|
| F |
F26 - Lower Level Codes
| F26 |
No Health Consequences or Impact |
13 |
|
| 13 |
30.2%
|
| F |
F05 - Lower Level Codes
| F05 |
Delay to Treatment/ Therapy |
11 |
|
| 11 |
25.6%
|
| F |
F01 - Lower Level Codes
| F01 |
Change in Therapeutic Response |
4 |
|
| 4 |
9.3%
|
| F |
F04 - Lower Level Codes
| 4 |
9.3%
|
| F |
F27 - Lower Level Codes
| F27 |
No Patient Involvement |
3 |
|
| 3 |
7.0%
|
| F |
F03 , F02 , F19 , F25 , F17 |
8
total:43 |
18.6%
|
| G |
G02 - Lower Level Codes
| G02 |
Electrical and Magnetic |
5 |
|
| G0201501 |
Electrode |
1 |
|
| G0202502 |
Patient Electrode |
3 |
|
| 9 |
28.1%
|
| G |
G03 - Lower Level Codes
| G03 |
Measurement |
3 |
|
| G03012 |
Sensor |
5 |
|
| 8 |
25.0%
|
| G |
G07 - Lower Level Codes
| G07 |
Others |
3 |
|
| G07001 |
Part/Component/Sub-assembly Term not Applicable |
2 |
|
| G07002 |
Appropriate Term/Code Not Available |
1 |
|
| 6 |
18.8%
|
| G |
G01 - Lower Level Codes
| G01 |
Biological and Chemical |
4 |
|
| 4 |
12.5%
|
| G |
G04 - Lower Level Codes
| 2 |
6.2%
|
| G |
G05 , G06 |
3
total:32 |
9.4%
|
|
EEG
|
Ju min Park , Republic of Korea
[ 2021-09-03 09:21:32.0]
|
|
What precautions should be taken in EEG research?
 [1] [1]
|
|
It's a real case!
|
Soo Jeong Choi , Republic of Korea
[ 2021-09-04 07:15:08.0]
|
|
Frequent case! User education is important.
 [1] [1]
|
|
Needs improving the usability
|
You Kyoung Lee , Republic of Korea
[ 2021-09-10 15:37:50.0]
|
|
No device problem was found, but the same problem happens, especially in older patients, according to nurse's experiences.
I think the manufacturer needs to consider improving the usability of the device.
 [1] [1]
|
|
device functionality
|
María Eugenia Villalobos-Hernández , (Non-APEC) Costa Rica
[ 2021-09-12 02:53:41.0]
|
|
In this case the device is working as expected, however in elder patients it was not working at all, so it is necessary to develop more research on the device use to elderly patient's skin conditions, because the situation happened before.
 [1] [1]
|
|
the gel
|
Pei-Hsuan Huang , The United States
[ 2021-09-14 04:42:39.0]
|
|
the gel should consider different situation
 [0] [0]
|
|
It was a useful case.
|
Soohyun Nam , Republic of Korea
[ 2021-09-16 10:15:12.0]
|
|
It was a useful case.
 [0] [0]
|
|
Anesthesia Deepness Brain Monitoring (ABDM) system & ABDM electrode sensor
|
Stevie Lee , Republic of Korea
[ 2021-09-16 13:43:12.0]
|
|
it's useful activity
 [0] [0]
|
|
About EEG
|
YoonSung Kim , Republic of Korea
[ 2021-09-16 17:49:11.0]
|
|
EEG signal is difficult, but this programe help to study about EEG.
 [0] [0]
|
|
device functionality in elder patients
|
Erika Masis , (Non-APEC) Costa Rica
[ 2021-09-20 10:03:57.0]
|
|
Because, in elder patients it was not working at all
 [1] [1]
|
|
device functionality in elder patients
|
Erika Masis , (Non-APEC) Costa Rica
[ 2021-09-20 10:52:10.0]
|
|
Because, in elder patients it was not working at all, more research should be done in older adults
 [1] [1]
|
|
Patient-Device Interaction Problem
|
Jieun Lee , Republic of Korea
[ 2021-09-20 21:28:41.0]
|
|
There should more research centered on electrode sensors effective for elderly patients, as the majority of patients with neurologic issues, thus requiring an EEG, are the population. Also, I am curious if there was an additional problem to the patient in this particular case, other than the delayed preparation for anesthesia.
 [0] [0]
|
|
EEG
|
Citra Bonnita Putri , Indonesia
[ 2021-09-21 00:55:31.0]
|
|
The activity is helpful.
 [0] [0]
|
|
Anesthesia Deepness Brain Monitoring (ABDM) system & ABDM electrode sensor
|
Regina Li , Hong Kong
[ 2021-09-21 11:19:47.0]
|
|
This is a very helpful activity.
 [1] [1]
|
|
Anesthesia Deepness Brain Monitoring (ABDM) system & ABDM electrode sensor
|
Seontae Kim , Republic of Korea
[ 2021-09-21 17:56:33.0]
|
|
Helpful activity
 [0] [0]
|
|



 [1]
[1]