|
Description
|
|
Patient Condition
- A 86-year-old male patient had an orthopedic surgery for femur fracture.
- The patient tried to apply the intermittent pneumatic compression device system to prevent deep vein thrombosis.
Events
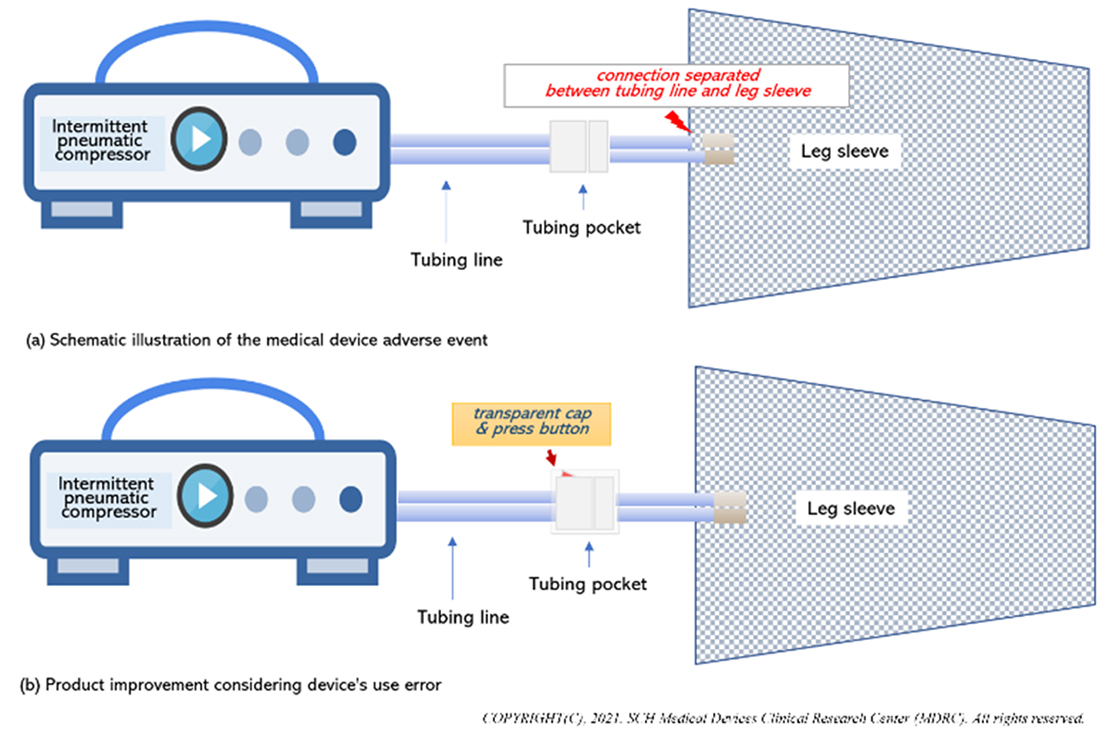
- The nurse found air leaks at the tubing line (connects leg sleeve and tubing pocket). Refer to Figure (a).
Post-event management & Health effect
- The patient applied a new leg sleeve.
- No significant health complications occurred.
Investigations (Cause, Improvements)
- Interview of patient carer
- Patients put the leg sleeve on or off repeatedly for their convenience in wards.
- During that process, the patient pulled off the leg sleeve repeatedly from the tubing pocket, without holding the tubing pocket, which resulted in a tear in the tube which connects the tubing pocket to the leg sleeve.
- Added a transparent cap on the tubing pocket, which the buttons on each side of the pocket need to be pressed for the leg sleeve to be disconnected.
- This prevents patients from pulling off the leg sleeve without holding the tubing pocket. Refer to Figure (b).
|
|
Click to Zoom
|
|
Annex
|
Code
|
#
|
%
|
| A |
A01 - Lower Level Codes
| A01 |
Patient Device Interaction Problem |
12 |
|
| A010102 |
Device Appears to Trigger Rejection |
1 |
|
|
13
|
28.9%
|
| A |
A05 - Lower Level Codes
| A05 |
Mechanical Problem |
3 |
|
| A0501 |
Detachment of Device or Device Component |
1 |
|
| A0504 |
Leak/Splash |
2 |
|
| A050402 |
Gas Leak |
3 |
|
|
9
|
20.0%
|
| A |
A12 - Lower Level Codes
| A12 |
Connection Problem |
2 |
|
| A1203 |
Disconnection |
2 |
|
| A1205 |
Loose or Intermittent Connection |
1 |
|
|
5
|
11.1%
|
| A |
A23 - Lower Level Codes
| A23 |
Use of Device Problem |
1 |
|
| A2301 |
Device Handling Problem |
1 |
|
| A2303 |
Improper or Incorrect Procedure or Method |
1 |
|
|
3
|
6.7%
|
| A |
A02 - Lower Level Codes
| A02 |
Manufacturing, Packaging or Shipping Problem |
3 |
|
|
3
|
6.7%
|
| A |
A22 , A26 , A25 , A03 , A04 , A11 , A14 , A15 , A21 |
12
total:45
|
26.7%
|
| B |
B13 - Lower Level Codes
| B13 |
Communication/Interviews |
13 |
|
|
13 |
39.4%
|
| B |
B01 - Lower Level Codes
| B01 |
Testing of Actual/Suspected Device |
8 |
|
|
8 |
24.2%
|
| B |
B03 - Lower Level Codes
| B03 |
Testing of Device from Same Lot/Batch Returned from User |
2 |
|
|
2 |
6.1%
|
| B |
B06 - Lower Level Codes
| B06 |
Testing of Model Variant |
2 |
|
|
2 |
6.1%
|
| B |
B15 - Lower Level Codes
| B15 |
Analysis of Data Provided by User/Third Party |
2 |
|
|
2 |
6.1%
|
| B |
B18 , B19 , B23 , B16 , B17 |
6
total:33 |
18.2%
|
| C |
C23 - Lower Level Codes
| C23 |
Usage Problem Identified |
13 |
|
| 13 |
41.9%
|
| C |
C07 - Lower Level Codes
| C07 |
Mechanical Problem Identified |
2 |
|
| C0703 |
Leakage/Seal |
3 |
|
| C0706 |
Stress Problem Identified |
1 |
|
| C070601 |
Deformation Problem |
1 |
|
| C070602 |
Fatigue Problem |
1 |
|
| 8 |
25.8%
|
| C |
C13 - Lower Level Codes
| C13 |
Operational Problem Identified |
3 |
|
| 3 |
9.7%
|
| C |
C19 - Lower Level Codes
| C19 |
No Device Problem Found |
1 |
|
| 1 |
3.2%
|
| C |
C01 - Lower Level Codes
| C010201 |
Endotoxin Contamination |
1 |
|
| 1 |
3.2%
|
| C |
C04 , C05 , C06 , C16 , C17 |
5
total:31 |
16.1%
|
| D |
D11 - Lower Level Codes
| D11 |
Cause Traced to User |
6 |
|
| D1101 |
Failure To Follow Instructions |
4 |
|
| D1102 |
Unintended Use Error Caused or Contributed to Event |
8 |
|
| 18 |
50.0%
|
| D |
D01 - Lower Level Codes
| D01 |
Cause Traced to Device Design |
2 |
|
| D0101 |
Design Inadequate for Purpose |
1 |
|
| D0102 |
Human Factors Engineering - Device Difficult to Operate |
2 |
|
| D0105 |
Missing or Inadequate Safety Measures |
1 |
|
| 6 |
16.7%
|
| D |
D08 - Lower Level Codes
| D08 |
Cause Traced to Training |
3 |
|
| 3 |
8.3%
|
| D |
D02 - Lower Level Codes
| D02 |
Cause Traced to Component Failure |
2 |
|
| 2 |
5.6%
|
| D |
D12 - Lower Level Codes
| D12 |
Known Inherent Risk of Device |
2 |
|
| 2 |
5.6%
|
| D |
D16 , D05 , D06 , D07 , D14 |
5
total:36 |
13.9%
|
| E |
E24 - Lower Level Codes
| E24 |
Others |
5 |
|
| E2403 |
No Clinical Signs, Symptoms or Conditions |
12 |
|
| 17 |
56.7%
|
| E |
E05 - Lower Level Codes
| E05 |
Vascular System |
2 |
|
| E0503 |
Embolism/Embolus |
1 |
|
| 3 |
10.0%
|
| E |
E21 - Lower Level Codes
| E21 |
Procedural Complications |
3 |
|
| 3 |
10.0%
|
| E |
E16 - Lower Level Codes
| E16 |
Musculoskeletal System |
1 |
|
| E1603 |
Bone Fracture(s) |
1 |
|
| 2 |
6.7%
|
| E |
E01 - Lower Level Codes
| 1 |
3.3%
|
| E |
E02 , E09 , E20 , E22 |
4
total:30 |
13.3%
|
| F |
F26 - Lower Level Codes
| F26 |
No Health Consequences or Impact |
15 |
|
| 15 |
51.7%
|
| F |
F05 - Lower Level Codes
| F05 |
Delay to Treatment/ Therapy |
5 |
|
| 5 |
17.2%
|
| F |
F10 - Lower Level Codes
| F10 |
Inadequate/Inappropriate Treatment or Diagnostic Exposure |
1 |
|
| F1001 |
Absence of Treatment |
1 |
|
| 2 |
6.9%
|
| F |
F27 - Lower Level Codes
| F27 |
No Patient Involvement |
2 |
|
| 2 |
6.9%
|
| F |
F08 - Lower Level Codes
| 1 |
3.4%
|
| F |
F09 , F21 , F24 , F01 |
4
total:29 |
13.8%
|
| G |
G04 - Lower Level Codes
| G04 |
Mechanical |
7 |
|
| G04002 |
Actuator |
1 |
|
| G04010 |
Balloon |
1 |
|
| G04034 |
Connector/Coupler |
7 |
|
| G04069 |
Hose |
1 |
|
| G04078 |
Joint |
1 |
|
| G04134 |
Tube |
4 |
|
| 22 |
66.7%
|
| G |
G06 - Lower Level Codes
| G06 |
Safety |
3 |
|
| G06004 |
Locking Mechanism |
1 |
|
| G06006 |
Safety Interlock |
1 |
|
| 5 |
15.2%
|
| G |
G01 - Lower Level Codes
| G01 |
Biological and Chemical |
3 |
|
| 3 |
9.1%
|
| G |
G05 - Lower Level Codes
| 1 |
3.0%
|
| G |
G07 - Lower Level Codes
| 1 |
3.0%
|
| G |
G03 |
1
total:33 |
3.0%
|
|
ballon? connector?
|
Soo Jeong Choi , Republic of Korea
[ 2021-09-04 07:32:25.0]
|
|
Component code is not easy for me.
 [2] [2]
|
|
Patient education
|
María Eugenia Villalobos-Hernández , (Non-APEC) Costa Rica
[ 2021-09-12 03:28:25.0]
|
|
It is very need to educate or train any patient in how to use the device or how to handle, this will contribute to prevent device misuses and therefore protect patient`s health. specially for elderly people or children.
 [1] [1]
|
|
It was a useful case.
|
Soohyun Nam , Republic of Korea
[ 2021-09-16 10:17:32.0]
|
|
It was a useful case. thank you.
 [0] [0]
|
|
Intermittent pneumatic compression device system & leg sleeve
|
Stevie Lee , Republic of Korea
[ 2021-09-16 14:03:05.0]
|
|
it was a useful activity
 [0] [0]
|
|
Incident vs adverse event
|
María Del Rocío Peinador Roldán , (Non-APEC) Costa Rica
[ 2021-09-16 23:22:21.0]
|
|
What's the difference between an incident and an adverse event?
 [0] [0]
|
|
Incident in intermittent pneumatic compression device system and leg sleeve
|
Erika Masis , (Non-APEC) Costa Rica
[ 2021-09-20 11:52:05.0]
|
|
Because of the lack of education of the patient in its use.
 [0] [0]
|
|
Incident in intermittent pneumatic compression device system and leg sleeve.
|
Erika Masis , (Non-APEC) Costa Rica
[ 2021-09-20 11:55:04.0]
|
|
Because of the lack of education of the patient in its use.
 [0] [0]
|
|
Intermittent pneumatic compression device system & leg sleeve
|
Seontae Kim , Republic of Korea
[ 2021-09-21 18:19:22.0]
|
|
Useful cases study
 [0] [0]
|
|



 [2]
[2]