IVD Medical Device Classification Tool
About This Tool
This tool is developed based on the "Principles of In Vitro Diagnostic (IVD) Medical Devices Classification (IMDRF/IVD WG/N64FINAL:2021)" to help the users in narrowing down the classification rules for an IVD medical device of interest.
Since there are multiple phrases with different meanings in some classification rules, we have added numbering system to identify the exact phrase of the rule e.g. Rule 1.1. The STEPs in this tool are developed by identifying the keywords for each phrase of the classification rules.
The user should fully understand the original IMDRF document before using this tool to be able to classify the IVD medical devices correctly because this tool focus only on the rules of the IMDRF document. There are other important sections in IMDRF document such as Definitions, General Principles, and Recommendations and Factors Influencing IVD Medical Device Classification, etc.
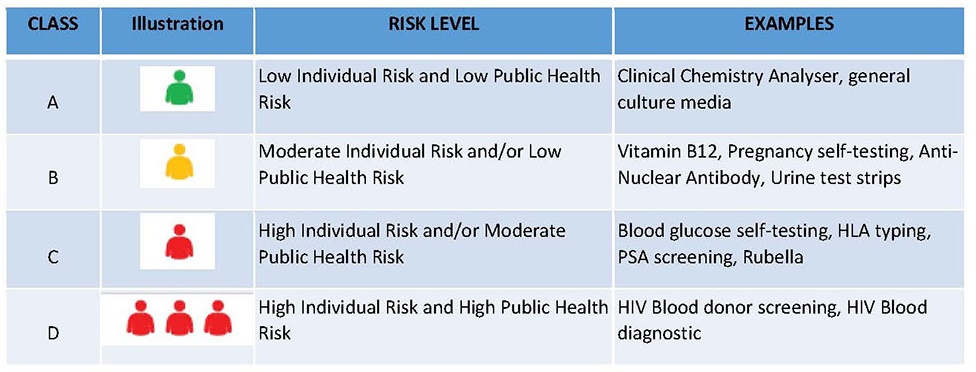
Figure 1 shows the general classification system and principle used in this tool. The idea of illustration is referenced from "Lubbers BR, Schilhabel A, Cobbaert CM, Gonzalez D, Dombrink I, Brüggemann M, Bitter WM, van Dongen JJ. The new EU regulation on in vitro diagnostic medical devices: implications and preparatory actions for diagnostic laboratories. Hemasphere. 2021 May;5(5)".
Figure 1: Proposed general classification system for IVD medical devices
How to Use
Start classifying the IVD medical device by selecting keywords in the STEP-1, STEP-2, and STEP-3. Multiple keywords for each STEP can be chosen. After STEP-3, the table below will show only the related rules for the IVD medical device being classified. The user can then decide which class the device should be put into.
The Determination of Device Class
The manufacturer should:
This tool is developed based on the "Principles of In Vitro Diagnostic (IVD) Medical Devices Classification (IMDRF/IVD WG/N64FINAL:2021)" to help the users in narrowing down the classification rules for an IVD medical device of interest.
Since there are multiple phrases with different meanings in some classification rules, we have added numbering system to identify the exact phrase of the rule e.g. Rule 1.1. The STEPs in this tool are developed by identifying the keywords for each phrase of the classification rules.
The user should fully understand the original IMDRF document before using this tool to be able to classify the IVD medical devices correctly because this tool focus only on the rules of the IMDRF document. There are other important sections in IMDRF document such as Definitions, General Principles, and Recommendations and Factors Influencing IVD Medical Device Classification, etc.
Figure 1 shows the general classification system and principle used in this tool. The idea of illustration is referenced from "Lubbers BR, Schilhabel A, Cobbaert CM, Gonzalez D, Dombrink I, Brüggemann M, Bitter WM, van Dongen JJ. The new EU regulation on in vitro diagnostic medical devices: implications and preparatory actions for diagnostic laboratories. Hemasphere. 2021 May;5(5)".
Figure 1: Proposed general classification system for IVD medical devices

How to Use
Start classifying the IVD medical device by selecting keywords in the STEP-1, STEP-2, and STEP-3. Multiple keywords for each STEP can be chosen. After STEP-3, the table below will show only the related rules for the IVD medical device being classified. The user can then decide which class the device should be put into.
The Determination of Device Class
The manufacturer should:
1. Take into consideration all the rules in order to establish the adequate classification for the device. Where an IVD medical device has multiple intended uses, as specified by the manufacturer, which can place the device into more than one class, it will be classified in the higher class.
2. Where more than one of the classification rules applies to the IVD medical device, it should be allocated to the highest class indicated, e.g. a self-testing for HIV would be a class D under rule 1 and not a class C under rule 4.
2. Where more than one of the classification rules applies to the IVD medical device, it should be allocated to the highest class indicated, e.g. a self-testing for HIV would be a class D under rule 1 and not a class C under rule 4.




